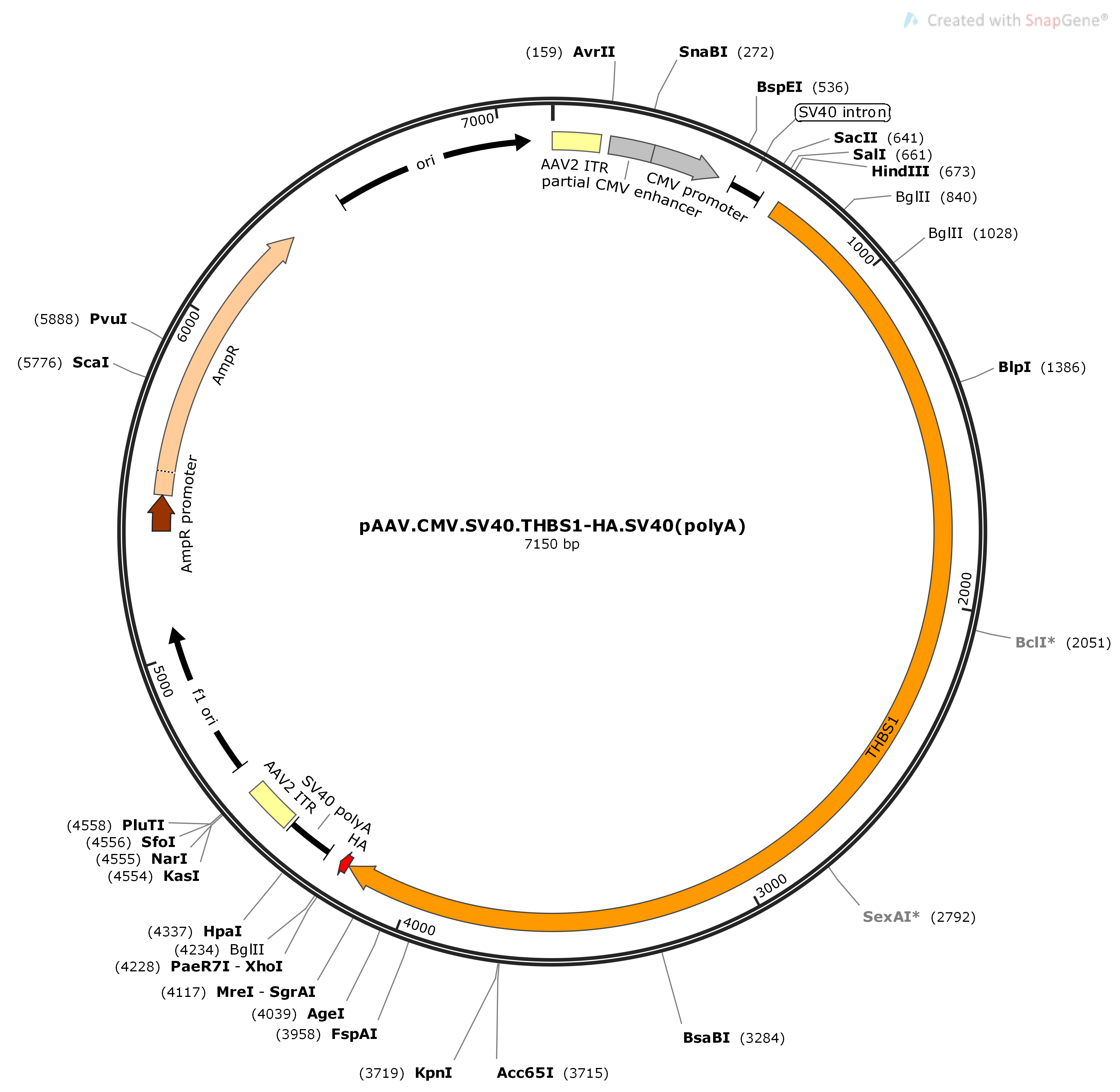
Constitutive promoters such as the human elongation factor 1α-subunit (EF1α), immediate-early cytomegalovirus (CMV), chicken β-actin (CBA) and its derivative CAG, the β glucuronidase (GUSB), or ubiquitin C (UBC) can be used to promote expression in most tissues ( Husain et al., 2009 Qin et al., 2010 Norrman et al., 2010). In some cases, such as those where a gene product is secreted, ubiquitous expression in all cell types is desired. Wang et al., 2008 Talbot et al., 2010 Katwal et al., 2013 Van Linthout et al., 2002 Cunningham et al., 2008 Klein et al., 2002 Ohlfest et al., 2005 Gray et al., 2011 Moreover, while the genome modifications are described in this review in the context of AAV and lentiviral vectors, they are certainly applicable to other vector systems.ĬBA (including derivatives: CAG, CBh, etc.) Although these are useful tools to control expression and/or cell specificity, they will not be discussed in this review. Other outstanding reviews are available for insulators ( Antoniou et al., 2013), self-complementary AAV ( McCarty, 2008), AAV serotype tropism ( Wu et al., 2006), retrovirus pseudotyping ( Matrai et al., 2010), and systems to induce/regulate expression using exogenously supplied trans-acting factors ( Toniatti et al., 2004). Lentivirus and AAV have been extensively reviewed elsewhere in the areas of their pros and cons, virology, uses, and development for gene transfer ( Nagabhushan Kalburgi et al., 2013 Kay et al., 2011 Grieger and Samulski, 2012 Segura et al., 2013). While keeping in mind size constraints, this review will discuss different cis-acting elements that have been engineered into lentivirus and AAV expression cassettes to enhance cell-specific transgene expression. 
Expression cassettes require thoughtful design due to foreign DNA packaging size constraints of AAV and lentivirus, approximately 4.1–4.9 kbs and 8–9 kbs, respectively ( Dong et al., 1996 Kumar et al., 2001). Transgene expression levels can be modulated by engineering the expression cassette to include the CMV enhancer (that includes transcription factor binding sites) or mRNA stability/nuclear export cis-acting elements (introns, polyA signals, or WPRE). 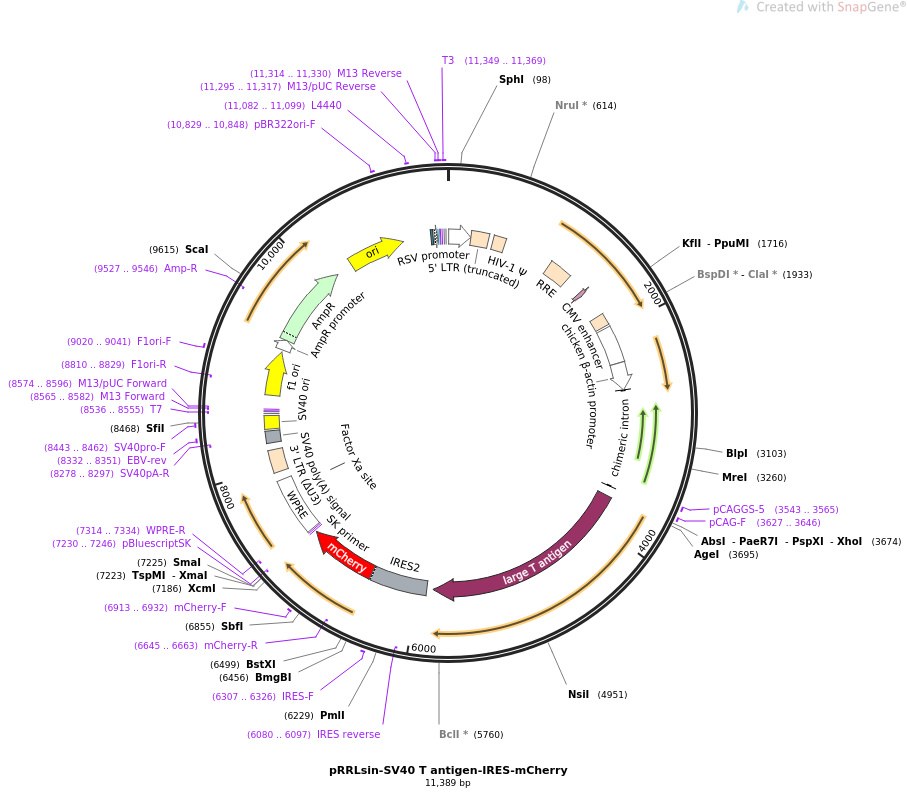
Target specificity can be honed by using cell-specific promoters or endogenous miRNAs. Lentivirus and AAV (adeno-associated virus) expression cassettes, prominently used in gene therapy, can be designed for target specificity and transgene expression levels ( Figure 1). Overexpression and non-targeted expression in some diseases, such as Rett Syndrome, is to be avoided ( Amir et al., 1999) however, in Hemophilia B, expression of Factor IX, a secreted protein present in the blood, is needed to be high and there is little concern of overexpression (reviewed in Cancio et al., 2013). Target specificity and an appropriate level of transgene expression can prevent unwanted phenotypes in other cells, an immune response, and possible toxicity. The cis-acting elements that regulate transgene expression can have as great of an impact on the success of gene therapy as the design of the vector capsid or envelope. In conclusion, this review can serve as a menu of vector genome design elements and their cost in terms of space to thoughtfully engineer viral vectors for gene therapy. All discussion bears in mind that expression cassettes have space constraints. Specifically, relevant studies indicating how cis-acting elements such as introns, WPRE, polyadenylation signals, and the CMV enhancer are highlighted to show their utility for enhancing transgene expression in gene therapy applications. The key differences relating to target specificity between ubiquitous and tissue-specific promoters are discussed, as well as how endogenous miRNAs and their target sequences have been used to restrict transgene expression. This review discusses how viral vector expression cassettes can be engineered with elements to enhance target specificity and increase transgene expression. For continued success, AAV and lentiviral expression cassettes need to be designed to meet each disease's specific needs. Over the last five years, the number of clinical trials involving AAV (adeno-associated virus) and lentiviral vectors continue to increase by about 150 trials each year.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
